 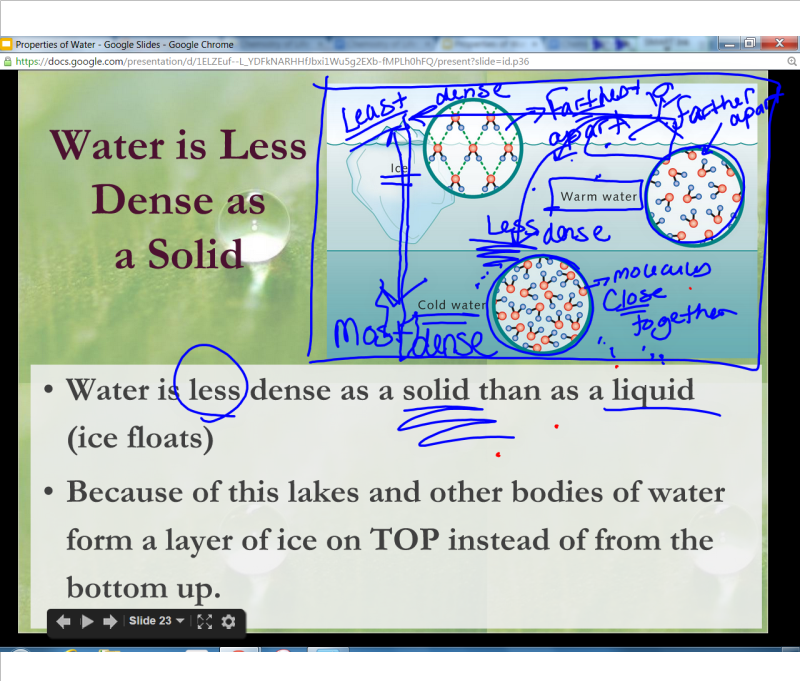
The force of crystallization of ice is capable of bursting water pipes and causes expansions of cracks in rocks, thus accelerating the erosion of mountains!Ī rough sketch of water molecules in ice crystal form is below. Have you or a family member (you wouldn't admit to this would you?) ever left a full container of water with a tight-fitting lid (or even a can of soda?) in the freezer? In other words, 10 cups of water put into the freezer is going to turn into 11 cups of ice when it freezes (oops). We are all aware that expansion of liquid water to ice exerts a tremendous force. Once the oceans filled with ice, life there would not be possible. If water did not expand when freezing, then it would be denser than liquid water when it froze therefore it would sink and fill lakes or the ocean from bottom to top. For example, fish in a pond survive the winter because ice forms on top of a pond (it floats) and effectively insulates (does not conduct heat from the pond to the atmosphere as efficiently) the rest of the pond below, preventing it from freezing from top to bottom (or bottom to top). Atoms can exist on their own or together as molecules. Matter is made up of small particles called atoms. This latter property is very important for organisms in the oceans and/or freshwater lakes. GCSE Edexcel Density of materials - Edexcel Required practical - how to calculate density. For this reason, ice floats on the water (like an ice cube in a glass of water). The density of water at 20☌ and 1 atm (standard atmospheric pressure) is approximately 998.21 kg/m³. In fact, its volume is a little over 9% greater (or density ca. Water expands when it freezes making it less dense than the water from which it freezes. Water, however, is unique in that it bonds in such a way that the molecules are held farther apart in the solid form (ice) than in the liquid. The density of the material is defined as the weight of the material per unit volume of it. For nearly every known chemical compound, the molecules are held closer together (bonded) in the solid state (e.g., in mineral form or ice) than in the liquid state. When water freezes, however, bonds are formed that lock the molecules in place in a regular (hexagonal) pattern.  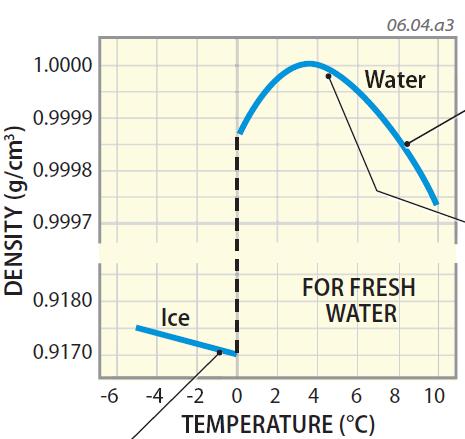
This effect also helps explain why you are supposed to add salt to ice when making ice cream or to add salt to water when cooking spaghetti (although, in this case, the effect on boiling point is minor and the added salt is mainly for flavor). Increasing salinity also lowers the temperature of maximum density. Normal seawater freezes at -2˚ C, 2˚ C colder than pure water. Incidentally, increasing salinity increases the boiling point and decreases the freezing point. As you add salts to seawater, you also change some other properties. The density of average seawater with a salinity of 35 o/oo (35 g/kg) and at 4˚ C is 1.028 g/cm3 as compared to 1.000g/cm3 for pure water. However, as dissolved solids are added to pure water to increase the salinity, the density increases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
